Electrochemistry
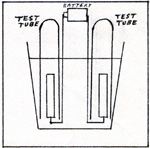
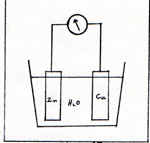
I perform an experiment in a beaker of water. The rest of the apparatus is a battery, two wires, two electrodes, and two test tubes. The electrodes can be carbon rods, pieces of platinum, or pieces of stainless steel. I arrange the wires and their electrodes in the beaker, while it is dry. (Fig. 1) |
Fig. 1
|
I pour water into the beaker. I fill two test tubes with water. I place the test tubes in the water of the beaker, and invert the tubes with their openings under water. The test tubes are still full of water. The electrodes are inside the test tubes. (Fig. 2) |
Fig. 2
|
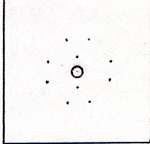
I add an ounce of sulfuric acid to a beaker of water. I connect the wires to a battery. Bubbles appear on the electrodes. Bubbles rise inside the test tubes. The gases that accumulate inside the test tubes, displace some of the water. The water level drops twice as fast in one test tube than in the other. I pick up the first test tube that fills up with gas. I quickly put a lit match to the mouth of the test tube. There is a minor explosion, nothing dangerous. The test tube contains hydrogen. I refill the test tube with water and replace it over the electrode. I pick up the other test tube, and quickly thrust a glowing wood splint into the tube. The wood bursts into flame. The test tube contains oxygen. It seems as if the electricity from the battery causes the water in the beaker to break apart into hydrogen and oxygen, two hydrogens for each oxygen. The sulfuric acid is regarded as just a conductor of electricity. It is much more complicated than that. The battery accepts electrons at its positive terminal, and issues electrons at its negative terminal. Any piece of metal that is connected only to the negative terminal gains a few electrons. It becomes slightly charged, negatively. When the negatively charged electrode is in contact with a dilute solution of sulfuric acid, it attracts protons. The protons happen to be part of H2OH+ ions. You see, the sulfuric acid reacts with water, and forms a positive ion and a negative ion. The protons leave the positive ions and settle on the negatively charged electrode. An electron leaves the metal electrode, and joins a proton. The combination of an electron and a proton is a hydrogen atom. Pairs of hydrogen atoms form hydrogen molecules. Any piece of metal that is in contact with the positive post of the battery, yields some electrons, which are drawn into the battery. This leaves the metal with a net positive charge. If the metal is in contact with a weak solution of sulfuric acid, it attracts sulfate ions, O2SO2-. A negative ion is pulled away from a sulfate ion by the positive electrode. A negative oxygen ion is shown in (Fig. 3) |
|
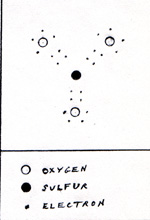
It has 8 protons and 10 electrons. The negative oxygen ion yields 2 electrons to the electrode. Thus the oxygen ion becomes an oxygen atom. Pairs of oxygen atoms form oxygen molecules. The remainder of the sulfate ion, after the departure of an oxygen ion, is a molecule of sulfur trioxide. (Fig. 4) |
|
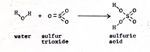
Sulfur trioxide reacts very strongly with water to form sulfuric acid. Although the sulfuric acid supplies all of the hydrogen and oxygen that issues from the two electrodes, in bubbles, there is no net consumption of sulfuric acid. For every pair of hydrogen atoms, and for each oxygen atom, produced in electrolysis, a water molecule disappears in the reaction. (Fig. 5) |
|
The effect of this step is to transfer the electrons of the hydrogen atoms to the sulfate ion. At first, the electrons reside in the water molecule. The water molecule combines with the sulfur trioxide. As soon as the sulfuric acid is constituted, it ionizes, leaving two electrons with the sulfate ion, as the protons move off. If we didn't think of all these details, it would appear to us that the only thing that happens is that water becomes separated into hydrogen and oxygen. I perform an experiment in which I measure an electric current. The instrument that measures current is a milliammeter. I convert the milliammeter into a meter that measures the rate of flow of electrons through a wire in units of electrons per second. The meter has connecting posts marked + and - . If I connect the meter to a battery correctly, I put - to - and + to + . Electrons move out of the negative post of the battery into the negative post of the meter, and out of the positve post of the meter into the positive post of the battery. Instead of connecting the wires to a battery, I connect the wire from the negative post of the meter to a small piece of zinc. I connect the wire from the positive post of the meter to a small piece of copper. I put the zinc and copper, as electrodes, into a beaker. (Fig. 6) |
|
I add distilled water to the beaker. The meter shows that there is a small current. Zinc ions are pulled into the water by the attraction of the negative side of the water molecules. For each Zn2+ that departs from the metal, two electrons remain on the metal. Shortly, the negative charge on the metal is large enough to attract Zn2+ ions as strongly as the water. As the zinc electrode accumulates electrons, the electrons destribute themselves so that they have as much space as possible between them without leaving a conductor. Some of the electrons move into the wire, thence into the meter. Electrons are conducted through the meter to the second wire, which carries electrons to the copper electrode. A water molecule that is in contact with the copper electrode, yields a proton. An electron from the copper electrode combines with the proton to form a hydrogen atom. |
|
The rest of the water molecule is a H-O- ion. The H-O- ion is repelled by the copper electrode, and attracted by the Zn2+ ions. Zn2+ ions migrate toward HO- ions. When the Zn2+ ions meet the HO- ions, they form the compound H-O-Zn-O-H, of which one molecule in a million remains in solution, while the rest precipitate as a crystal. There is no complete migration of ions from one electrode to another electrode. The meter reads a current of 1013 electrons per second. That is a very small current. It takes a current of about 1018 electrons per second to make a flashlight bulb glow dimly. A practical cell for producing electric current needs more ions in solution. I add a switch to the circuit. Switching has the effect of connecting or disconnecting a wire. The meter responds instantaneously to the switching. No matter how long I make the wire, the response is just as fast. This does not prove anything about the speed of an electron in the wire. Electrons in a wire have speeds between zero and 106 cm/sec, depending on the temperature. The heat motion of the electron does nothing to advance the electron along the wire. The electron retreats as often as it advances. The signal, or the information that a current exists, moves at close to the speed of light. The electron in a wire always dawdles. The motion due to an electron circuit is usually slower than 1cm/sec. Current is the net number of electrons that pass a point in the wire in one second, going in the same direction. It does not measure the speed of the electrons. When a zinc atom is in place on the surface of a metal, its negative charge and positive charge are equal. The free electrons on the surface of a metal are so distributed as to have no net force on an electron. I am imagining electrons that are standing still. A zinc ion, Zn2+ departs from the metal at point X. (Fig. 7) |
Fig. 7
|
Suddenly, the density of negative charge is increased by two electrons at point x , where the Zn2+ leaves them behind. With the Zn2+ gone, there is a push away from point x. All of the electrons on the surface of the zinc, move until they are once more spaced so that the net force on each electron is zero. The adjustment takes very little time. I imagine a slow motion picture of the process. The electrons closest to the center move first, because the field is strongest at the center, Each electron moves along a radius from that center. (Fig. 8) |
Fig. 8
|
Electron C is at the center. What looks like ripples is pairs of bits that are emitted by electrons constantly, but randomly in all directions. The pairs move at the speed of light. Bit pairs from electron c reach electron e sooner than bit pairs from electron c reach electron f. When electron e receives bits from electron c, electron e starts to move a little closer to electron f. Therefore electron f receives bits from both c and e. As each electron is hit by more bits from one direction, it starts to move. That motion is slow, although the bit pairs move at the speed of light, carrying information at that speed. When I connect a wire to the zinc electrode, the electrons throughout the wire shift to accommodate the extra electrons that are in the zinc. In a complete circuit, in which electrons can pasds continuously through the wire, the opening or closing of a switch affects the most distant electron at the speed of light. The cause of all the motion of electrons and ions in this experiment is the difference between copper and zinc. Copper and zinc are closely related, because copper has 29 protons, and zinc has 30 protons. (Fig. 9) |
Fig. 9
|
The 30 protons of the zinc nucleus pull the electrons closer to the center of the atom, than the 29 protons of copper. Each layer of electrons in the zinc atom is more densely crowded than the corresponding layer in the copper atom. As a result, the first 28 electrons of the zinc atom are more effective in shielding the nuclear charge. The outermost two electrons of the zinc atom are more loosely held than the outermost single electron of the copper atom. The third layer of electrons in the copper atom has 18 electrons that are not as tightly held as the third layer of electrons in the zinc atom. Although it is harder to separate the outermost electron plus one of the third layer electrons from the copper atom, when sufficient energy is transferred to these two electrons, both of them leave the atom. A copper atom has a double charge. Water attracts Cu2+ as strongly as it attracts Zn2+. The difference is that Cu2+ can't easily break away from the copper metal, but Zn2+ can easily break away from zinc metal. That difference is what drives the electrochemical cell. An electric cell that can produce enough current to light a flashlight bulb, can be set up in two beakers with a narrow tube between them. (Fig. 10) |
Fig. 10
|
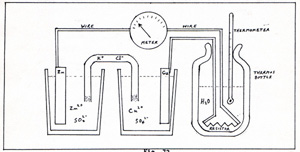
The tube is shaped like an inverted U. It contains a solution of K+ and Cl-. Cotton plugs keep the solution in the tube from mixing too rapidly with the solutions in the beakers. The beaker with the zinc electrode contains a solution of 16 grams of zinc sulfate in 100 grams of water. The beaker with the copper electrode contains a solution of 16 grams of copper sulfate in 100 grams of water. The advantage of this cell is the presence of copper ions in the solution surrounding the copper electrode. There is no necessity to tear water molecules apart to supply ions. While the reaction is in progress, copper atoms settle on the surface of the copper electrode. The KCl bridge completes the circuit, so that zinc ions have a place to migrate to. Otherwise, the zinc ions clutter up the immediate vicinity of the zinc electrode, and prevent other zinc ions from leaving the metal. Instead of the light bulb, I connect to the wires, a sensitive voltmeter that draws little or no current. I replace the scale of the voltmeter with another scale that gives direct readings in ergs per electron. It tells me how much potential energy an electron has when it leaves the zinc electrode. That is potential energy in the electron-copper electrode system. It gives a reading of 1.76 x 10-12 erg per electron. If the wire which connects the electrodes were a perfect conductor, the electron would arrive at the copper electrode with a kinetic energy of 1.76 x 10-12 erg. A perfect conductor does not intercept any of the energy. A less than perfect conductor accepts energy from the migratory electrons. When an electron interacts with any part of an atom, energy is transferred. On the average, energy passes from the particle with the higher temperature to the particle with the lower temperature. Interactions between electrons and atoms, slow down the conducting electrons , and raise the temperature of the atoms. This amounts to a conversion of electrical energy to heat energy. The wire is a less-than-perfect conductor because it intercepts some of the energy of the conducting electron. I dislike using such customsry expressions as " conversion of energy to other forms of energy " . Be warned that energy remains a product of the imagination, no matter how much it sounds like a real thing. I prefer to think of charge as the basic substance. I don't offer a detailed mechanism for each transfer of energy in terms of transfer of charge, because it sounds monotonous. I connect both, light bulb and meter, to the same wires. In a sense, I am measuring the energy per electron across the light bulb. Each electron must pass through the light bulb, having yielded that much energy to the bulb as heat and light. (Fig. 11) |
Fig. 11
|
The meter reads much less with the bulb in the circuit than without the bulb. The energy per electron is reduced from 1.76 x 10-12 to about half that. The problem is not in the bulb, but in the cell. As long as the switch is off, the electrodes are in equilibrium with the solution. The zinc electrode has its maximum concentration of negative charge. Less charge would cause Zn2+ to leave. More charge would cause Zn2+ to return. The copper electrode has a very slight negative charge, very close to zero. The Cu2+ has very little tendency to leave its electrons. If the charge were more negative, Cu2+ would return from the solution. If the charge were less negative, more Cu2+ would leave the electrode. |
|
The meter reading of 1.76 x 10-12 erg per electron that I get when the bulb is not in the circuit, is due to the equilibrium densities of electrons on the electrodes. Between the zinc electrode and the copper electrode, imagine a conductor that can pass only one electron. The electron is propelled by many excess electrons in the zinc electrode, and repelled in the opposite direction by very few excess electrons in the copper electrode. The potential energy , as the electron departs from the zinc, is 1.76 x 10-12 erg. When the bulb is in the circuit, electrons leave the zinc electrode faster than Zn2+ can depart from the zinc electrode, and electrons enter the copper electrode faster than Cu2+ ions can move toward the copper electrode. Therefore, the density of excess electrons on the zinc electrode is diminished, and the density of excess electrons on the copper electrode is increased. Each electron that leaves the zinc ion under those circumstances, is pushed by fewer electrons on the zinc electrode, and is repelled in the opposite direction by more electrons on the copper electrode. Therefore, the potential energy per electron is less than 1.76 x 10-12 erg, when the bulb is in the circuit. The electron that leaves the zinc electrode when the current is high, has less energy than the electron that leaves the zinc electrode, when the current is low. The Zn2+ then arrives at the water molecule with greater energy than when there is no current. The Cu2+ ion arrives at the copper electrode with more energy, when the current is high, because there is more excess negative charge on the copper electrode. The energy of the electron is variable, and the energy of a positive ion is variable. Both depend on the current. Regardless of the current, the total energy of two electrons, one Zn2+, and one Cu2+ is constant. It is the energy of the reaction: Zn + Cu2+ ----->Cu + Zn2+ zinc + copper sulfate -----> copper + zinc sulfate Whether a reaction is carried out fast or slowly, or electrolytically, or directly, the energy of the reaction is always the same. I conduct an experiment, using the electric cell and a thermos bottle, a resistor, and a themometer. (Fig. 12) |
Fig. 12
|
The meter measures current in electrons per second. The resistor is a less-than-perfect conductor. The lower the conductance, the higher the resistance. I use a 55 ohm resistor. Its purpose is to prevent too rapid progress of the reaction. When the potential energy per electron is 10-10 erg, the 55 ohm resistor restricts the current to 7.1 x 1018 electrons per second. I place the resistor in a thermos bottle. I add 40 cm3 of water. I put a thermometer in, to observe the temperature of the water. I keep a record of the time and temperatue. At the start of the experiment, the temperature of the water is 298 K. The meter reads 1.25 x 1017 electrons per second. I let the experiment run for 24 hours. I collect the following data:
The total energy transacted in this experiment is 1.9 x 1010 erg. (8.64 x 104 sec) x (1.25 x 1017 elec/sec) x (1.76 x 10-12 erg/elec) = (1.9 x 1010 erg) A good procedure, when solving such a problem, is to operate on the units separately: (sec) x (elec/sec) x (erg/elec) = (erg) A sec in the numerator cancels a sec in the denominator. An elec in the numerator cancels an elec in the denominator. The only unit that is not cancelled is the erg, the unit of energy. When one gram of water absorbs 4.185 x 107 erg of energy, its temperature rises 1 degree. In this experiment, 40 grams of water have an increase of temperature of 11.3o. (11.3 deg) x (40 gram) x (4.185 x 107 erg/deg gram) = (1.9 x 1010 erg) The energy comes out the same, whether it is calculated by electrical data, or by quantity of heat. The experiment confirms the observation that the electrical energy that this cell yields, when the current is minimal, is 1.76 x 10-12 erg per electron. I want to compare this with the total energy of the reaction between zinc and copper sulfate. |
|
I have a beaker of 200 grams of water, in which I dissolve 15.9 grams of dehydrated, pulverized copper sulfate. I let the solution rest until it is at room temperature, 298 K. I pour the solution into a thermos bottle. I place a thermometer in the solution. I toss in about ten small pieces of zinc, having a total mass of 6.54 grams. In a few minutes, the zinc disappears. The thermometer reaches its maximum reading of 324 K. The water absorbs: (26 deg) x (4.185 x 107 erg/g deg) x (200 g) = 2.18 x 1011 erg) The number of atoms of zinc in 6.54 grams of zinc is 6.022 x 1022. There are two electrons for every Zn2+ ion. Therefore, this reaction involves 2 x 6.022 x 1022 = 1.2044 x 1023 electrons. 2.18 x 1011 erg/ 1.2044 x 1023 elec = 1.81 x 10-12 erg/elec The total energy of the reaction is 1.81 x 10-12 erg per electron. The maximum electrical energy to be derived from this cell is 1.76 x 10-12 erg per electron. 1.81 x 10-12 - 1.76 x 10-12 = 5 x 10-14 The minimum heat energy to develop within the cell is 5 x 10-14 erg per electron. If I connect a perfect conductor between the electrodes, the heat within the cell becomes 1.8 x 10-12 erg per electron. Of no importance, but of some interest, is the fact that the copper sulfate powder is white. In solution it is blue. The zinc starts out metalic, and then disappears. The blue color leaves the solution, which turns colorless. The bottom of the vessel becomes covered with bits of red copper. People are still disappointed with this sort of outcome for every process whose purpose it is to supply energy. In every case, some of the energy must be in the form of heat, and some of the heat must pass directly to the surroundings, without any benefit to people. If there were only one process that could yield more energy than it consumes, the excess energy could be used to produce more of the raw material that goes into the process.Then the process would drive itself, and make a profit, too. That would be an example of perpetual motion. Unfortunately, this is quite impossible. We know that this is impossible, from experience, and all existing theories. |
Other Essays | ||||||
|
|
|
|
|
|
| |
|
|
|
|
|
| |
|
|
||
| previous page |
|
|
|
|
||
|
|
||